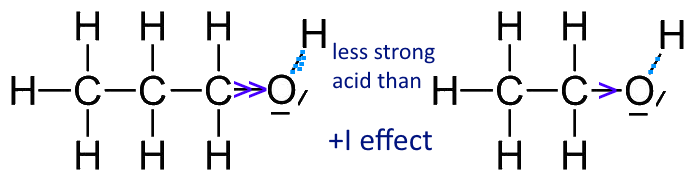
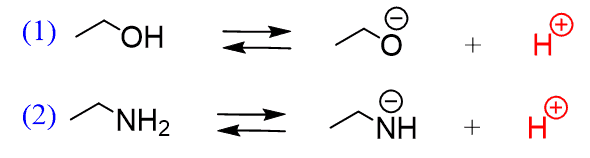
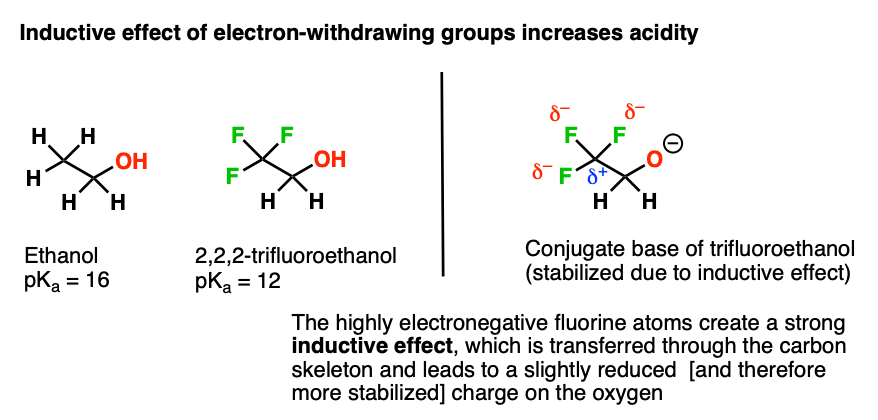
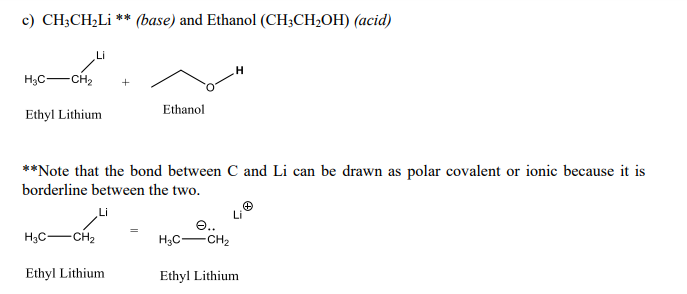
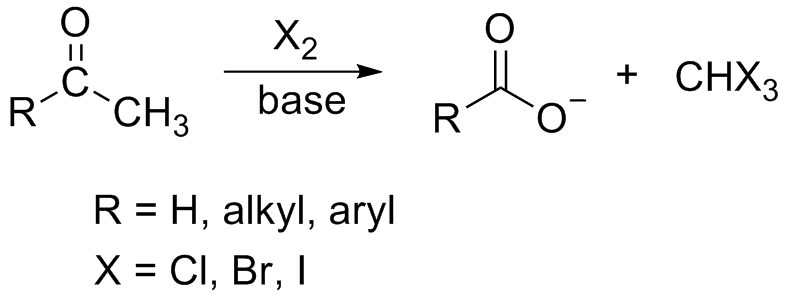
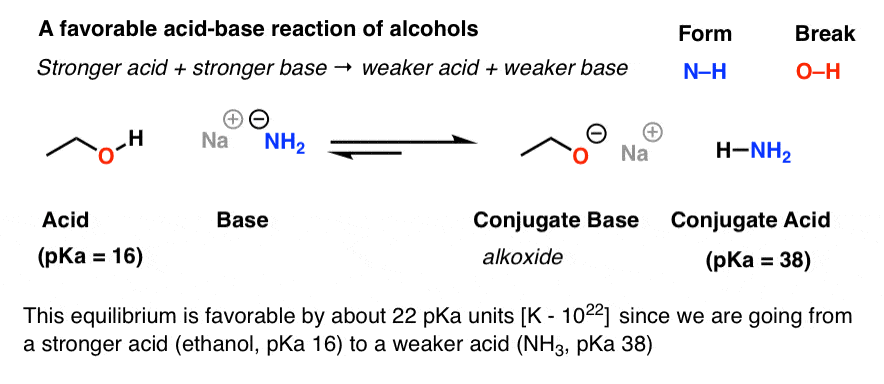Draw the Lewis structure for the conjugate base from the reaction of ethanol with a generic base. Include all lone pairs of electrons and any nonzero formal charges. | Homework.Study.com

Ethanol condensation to butanol at high temperatures over a basic heterogeneous catalyst: How relevant is acetaldehyde self-aldolization? - ScienceDirect

Valorization of Ethanol: Ruthenium-Catalyzed Guerbet and Sequential Functionalization Processes | ACS Catalysis

Kante Concrete Tabletop Fire Pit with 6" Light Gray Base, Ethanol Fire Pit for Indoor&Outdoor, Portable Rubbing Alcohol Tabletop Fire Bowl, Mini Fireplaces for Smores Maker (Basic)

Northlight 10.5 in. Bio Ethanol Round Portable Tabletop Fireplace with White Base 34808726 - The Home Depot
Mechanism of reaction between caprolactam and ethanol in the presence... | Download Scientific Diagram
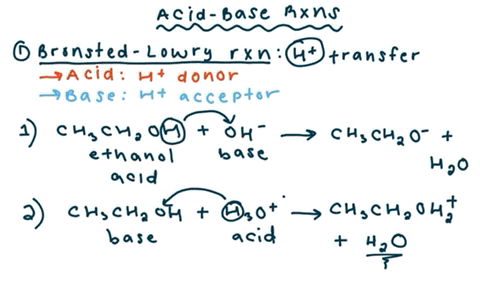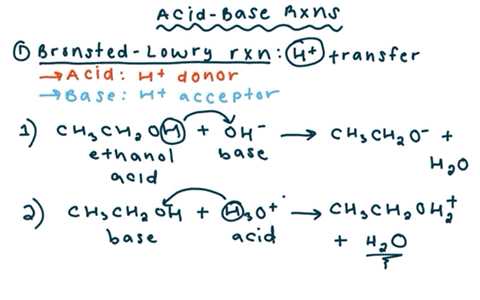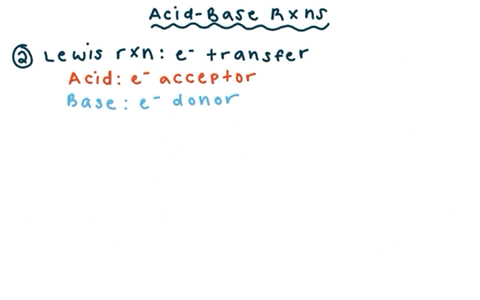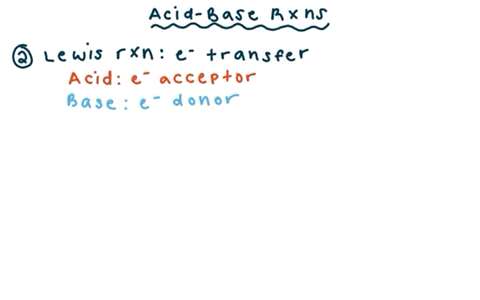
SOLVED: reaction belween butylithium and ethanol: Label each reactant in this reaction as Bronsted Consider the acid or base. butyllithium ethanol butane Ilthium ethoxide CH,Li C,H;OH C,H,o LiOC,Hs acid base acid base

Why is the conjugate acid of an ether/alcohol more acidic than that of a hydronium ion? : r/chemhelp